Earlier Attempts At The Classification Of Elements
There have been several attempts to classify the elements based on their properties. The first attempt to classify elements resulted in the classification of metals and non-metals from then-known elements.
Döbereiner’s Triads (1817)
Johann Wolfgang Dobereiner (a German chemist) made an effort in 1817 to organise elements with similar properties into groups of three elements each. He gave them the name ‘triads’.
Dobereiner demonstrated that the middle element’s atomic mass was approximately the average of the other two elements’ atomic masses when the three elements in a triad were arranged in increasing order of atomic masses.
Importance: It is extremely important in estimating the atomic mass and properties of the middle element. These elements resemble their properties even to this day.
Limitation: The total number of elements were more than that of those encompassed in Dobereiner’s Triad. From the elements known at the time, Dobereiner was able to identify these three triads: Li, Na, K; Ca, Sr, Ba; Cl, Br, I.
Newlands’ Law Of Octaves (1866)
In 1866, an English scientist named John Newlands grouped the known elements in order of increasing atomic weights and discovered that every eighth element had properties close to the first.
He compared it to musical octaves i.e. sa, re, ga, ma, pa, da, and ni or known as do, re, mi, fa, so, la, ti in the west. And thus, it was named “Law of Octaves.”
A part of the original form of Newlands’ octaves is shown in the table below.
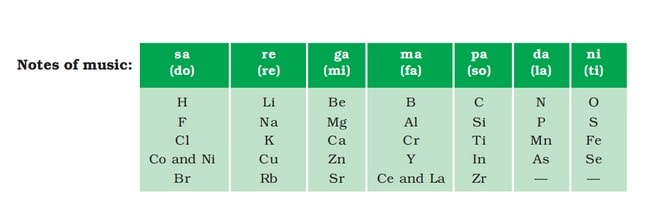 Limitations: This law could be applied only upto calcium. Every eight element, after calcium, do not possess the same properties as the first
Limitations: This law could be applied only upto calcium. Every eight element, after calcium, do not possess the same properties as the first
Newland assumed that nature had only 56 elements and neglected the possibilities of any future discoveries. Later on, however, several additional elements were found with properties that defied the Law of Octaves.
Newlands altered two elements in the same slot and put several unlike elements under the same column. Cobalt and nickel, for example, are in the same slot and in the same column as fluorine, chlorine, and bromine, which do not have similar properties.
Iron, which possesses properties similar to cobalt and nickel, has been separated from these metals. As a result, Newlands’ Law of Octaves only applied to lighter elements.
Mendeleev’s Periodic Table (1834-1907)
Dmitri lvanovich Mendeleev, a Russian chemist, was the most important contributor for the early development of a periodic table of elements wherein the elements were arranged on the basis of their fundamental property, the atomic mass and also on the similarity of chemical properties. Only 63 elements were known at his time.
To classify these elements:
- He examined the relationship between the atomic masses of the elements and their physical and chemical properties.
- Among chemical properties, he concentrated on the compounds formed by the elements with oxygen and hydrogen as they are very reactive and form compounds with most elements.
- He took the formulae of the hydrides and oxides formed by an element as one of the basic properties of an element for its classification.
- He then arranged 63 elements in the increasing order of their atomic masses and found chat there was a periodic recurrence of elements with similar physical and chemical properties.
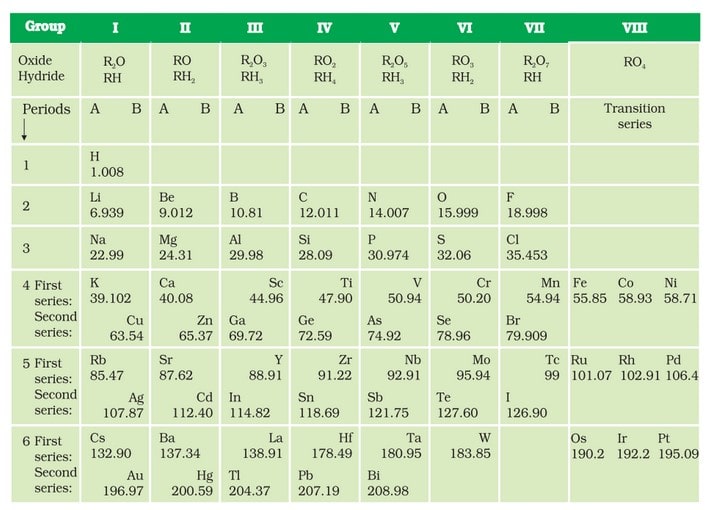
He observed that elements with similar properties fall in the same vertical column. These vertical column are called groups and horizontal rows of elements are called periods. - On this basis, Mendeleev gave a Periodic Law, which states that ‘the properties of elements are the periodic function of their atomic masses’.
Features Of Mendeleev’s Periodic Table
The important features of this table are :
- This table contains 8 vertical columns, called groups and 6 horizontal rows, called periods.
- In his table, Mendeleev’s left gaps for the elements not discovered at that time. He named such elements by prefixing a Sanskrit numeral Eka (one), divi (two), tri (three) etc., to the name of the preceding similar (analogous) element in the same group.
e.g. Eka-boron, Eka-aluminium, Eka-silicon, which after their discovery were named as scandium, gallium and germanium respectively. - He also predicted the atomic masses and properties of several elements that were not known at that time. Elements like scandium, gallium and germanium have properties similar to those predicted by Mendeleev.
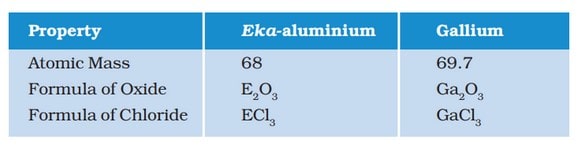
The properties of Eka-aluminium predicted by Mendeleev and those of element, gallium which was discovered later that replaced Eka-aluminium are listed as follows - In order to group the elements having similar properties together, at some places, Mendeleev had to place an element with a slightly greater atomic mass before an element with a slightly lower atomic mass.
- Noble gases like helium (He), neon (Ne), argon (Ar) etc., were discovered very late because they are very inert and present in extremely low concentrations in our atmosphere. One of the strengths of Mendeleev’s periodic table was that, when these gases were discovered, they could be placed in a new group without disturbing the existing order.
Limitations Of Mendeleev’s Periodic Table
Although this table was greatly helpful for the study of elements but a few anomalies could not be explained on the basis of this table. These anomalies are:
- Position of Hydrogen
- Position of Isotopes : Isotopes of an elements have similar chemical properties but different atomic masses. In Mendeleev’s periodic table, no place was given to these elements, e.g. the element chlorine has two isotopes, Cl-35 and Cl-37, having atomic masses of 35 and 37 respectively.
The placing of these two isotopes of chlorine (having different atomic masses) in the same group of periodic table could not be explained by Mendeleev’s periodic law.
- Uncertainty in Atomic Masses : Another problem was that the atomic masses do not increase in a regular manner in going from one element to the next.
So, it was not possible to predict how many elements could be discovered between two elements especially when we consider the heavier elements.
- Placing of Heavier Element before the Lighter One : Few elements, those possess higher atomic mass were placed before elements having a lower atomic mass.
e.g. Argon (39.9) was placed before potassium (39.1), cobalt (58.9) before nickel (58.7), tellurium (127.60) before iodine (126.9).
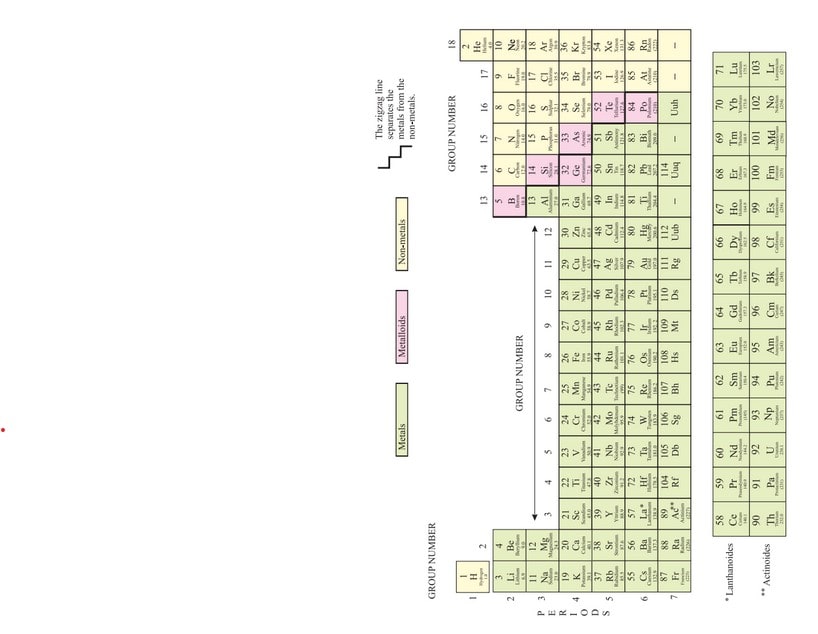
Modern Periodic Table
In 1913, Henry Moseley showed that the atomic number of an element is a more fundamental property. On the basis of this, he modified Mendeleev’s Periodic Law as “physical and chemical properties of the elements are a periodic function of their atomic number” .
This is called Modern Periodic Law. When the elements were arranged in the increasing order of their atomic number, the obtained table is called Modern Periodic Table.
Features Of Modern Periodic Table
This table has 18 vertical columns, known as groups and 7 horizontal rows, known as periods.
Features of Groups
A few important features of the elements present in groups are as follows:
- The groups are not divided into sub-groups. The elements present in a group have the same number of valence electrons.
- The elements present in a group have the same valency.
- The number of shells increases as we go down the group.
- The elements present in a group have identical chemical properties.
- The physical properties of the elements such as melting point, boiling point, density in a group vary gradually.
Features of Periods
The important features of the elements in a period are as follows:
- Elements of a period do not have the same number of valence electrons but they contain the same number of shells.
- The number of valence shell electrons increases by one unit as the atomic number increases by one unit on moving from left to right in a period. Therefore, the atoms of different elements with same number of shells are placed in the same period.
- As the number of valence shell electrons change, the chemical properties of the elements also change.
- Different periods have different number of electrons which can be explained on the basis of filling of electrons into various shells.
We can explain the number of elements in periods is based on how electrons are filled into various shells.
Maximum number of electrons that can be accommodated in a shell is given by the formula 2n²
Where, n = number of given shell from the nucleus.
e.g.
- K Shell – 2 x (1)² = 2, hence the first period has 2 elements and is called very short period.
- L Shell – 2 X (2)² = 8, hence the second period has 8 elements and is called short period.
- M Shell – 2 x (3)² = 18, but the outermost shell can have only 8 electrons, so the third period also has only 8 elements thus, it is also called short period. Fourth and fifth periods have 18 elements and are called long periods.
- Sixth and seventh periods have 32 elements and are also called long periods.
Position Of The Elements In The Modern Periodic Table
In order to find the position of an element in the periodic table, first write its electronic configuration and then find period and group number from its electronic configuration in the following way:
- The period number of an element is equal to the number of electron shells in its atom.
e.g. if the atom of an element has 2 electron shells (K and L), then it belongs to 2nd period. - If two (or more) elements have the same number of valence shells, then they belong to the same period of the periodic table.
- The group number of en element having upto two valence electrons is equal to the number of valence electrons.
e.g. if an element has 1 valence electron, it belongs to group 1. And if an element has 2 valence electrons, it belongs to group 2 of the periodic table. - The ‘group number’ of an element having more than 2 valence electrons is equal to the number of valence electrons plus 10′.
e.g. if an element has 3 valence electrons then its group number will be 3 + 10 = 13. - If two (or more) elements have the same number of valence electrons then they belong to the same group of the periodic table.
Explanation Of The Anomalies Of Mendeleev’s Periodic Table
Different anomalies of Mendeleev’s periodic table can be explained with the help of modern periodic cable as:
- The fundamental basis for modern periodic table is atomic number, not atomic mass, hence it is more accurate.
- Since, the table is based on atomic number and isotopes have same atomic number and chemical properties, so they can be put at one place in the same group of the periodic table.
- In this periodic table, a unique position has been given to hydrogen. It is kept at the top left corner because of its unique characteristics.
- The position of cobalt and nickel is justified itself because atomic number of cobalt is less than atomic number of nickel.
Trends In The Modern Periodic Table
In this table, some properties show a regular trend when we move along a period from left to right or in a group from top to bottom. These properties are:
VALENCY
It is the combining capacity of an atom of an element to acquire noble gas configuration. It depends upon the number of valence electrons present in the outermost shell of its atom.
For the elements of group 1, 2, 13 and 14, valency = number of valence electron(s), whereas for the elements of group 15 onwards, valency= 8 – valence electrons. Thus, the valencies of elements of different groups are as follows:
Valency of group 1 elements is 1;
Valency of group 2 elements is 2;
Valency of group 13 elements is 3;
Valency of group 14 elements is 4;
Valency of group I5 elements is 3;
Valency of group 16 elements is 2;
Valency of group 17 elements is 1;
Valency of group 18 elements is 0.
Variation Along a Group
In a group, outer electronic configuration is same for all the elements, so all have the same number of valence electrons and the valency. e.g. all the elements of group 1 have valency= 1.
| Group 1 Elements | Symbol | Electronic Configuration | No.of Valence Electron |
| Lithium | Li | 2,1 | 1 |
| Sodium | Na | 2,8,1 | 1 |
| potassium | K | 2,8,8,1 | 1 |
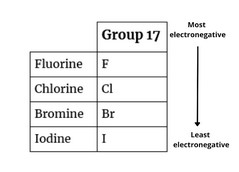
Similarly, for the elements of group 17 , valency = 1 (8 – 7 = 1).
| Group 1 Elements | Symbol | Electronic Configuration | No.of Valence Electron |
| Fluorine | F | 2,7 | 7 |
| Chlorine | Cl | 2,8,7 | 7 |
| Bromine | Br | 2,8,18,7 | 7 |
| Iodine | I | 2,8,18,18,7 | 7 |
Variation Along a Period
The valency increases from 1 to 4 (till group 14) and then decreases to zero from group 15 to 18.
| Group | 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 |
| Valency | 1 | 2 | 3 | 4 | 3 | 2 | 1 | 0 |
ATOMIC SIZE
It refers to the radius of an atom. It may be visualised as the distance between the centre of the nucleus and the outermost shell of an isolated atom. It is measured in picometres: [1 pm (picometre) = 10⁻¹²m].
e.g. atomic radius of hydrogen atom is 37 pm (or 37 x 10⁻¹²m)
Variation Along a Group
The atomic size increases down the group. This is because new shells are being added as we go down the group. This increases the distance between the outermost electrons and the nucleus. As a result, the atomic size increases inspite of the increase in nuclear charge.
| Group 1 | Atomic radii (pm) |
| Li (Lithium) | 152 |
| Na (Sodium) | 186 |
| K (Potassium) | 231 |
| Rb (Rubidium) | 244 |
| Cs (Caesium) | 262 |
| Fr (francium) | 270 |
Smallest atom to Biggest atom
Variation Along a Period
The atomic radius decreases in moving from left to right along a period. This is due to an increase in nuclear charge which tends to pull the valence electrons closer to the nucleus and reduces the size of the atom. e.g.
| Third period elements | Na | Mg | Al | Si | P | S | Cl |
| Atomic radii (pm) | 186 | 160 | 143 | 118 | 110 | 104 | 99 |
Size of atoms decreases
Metallic and Non-metallic Properties
Elements having a tendency to lose one or more electrons and form positive ions are called metals. These are present on the left side as well as in the centre of the periodic table. The tendency of these elements to lose electrons is called their metallic character.
Because of the formation of positive ions, these are also called electropositive elements. Non-metals are the elements which have a tendency to gain one or more electrons to form negative ions. Thus, these are electronegative elements.
These are present on the right side in the periodic table. The non-metallic character of the elements is due to their electron accepting tendency. There are some elements which exhibit the properties of both metals and non-metals. These are called metalloids. In the modern periodic table, a zig-zag line separates metals from non-metals.
The borderline elements-boron, silicon, germanium, arsenic, antimony, tellurium and polonium are intermediate in properties so they are called metalloids or semi-metals.
Variation Along a Period and a Group
As the effective nuclear charge acting on the valence shell electrons increases across a period, the tendency to lose electrons will decrease.
Down the group, the effective nuclear charge experienced by valence electrons decreases because the outermost electrons are farther away from the nucleus.
Therefore, these can be lost easily. Hence, metallic character decreases across a period and increases down a group. Non-metallic character, however increases across a period and decreases down a group.
e.g. in case of elements of third period, metallic and non-metallic character vary in the following manner.
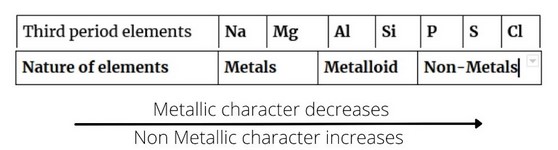
However, in groups, the order of variation of metallic and non-metallic character is as follows:

Nature Of Oxides
Oxides of the metals are of basic nature while those of non-metals are acidic. This means that along a period, the basic character of the oxides of the elements decreases while their acidic character increases
| oxide | Name | Nature |
| Na₂O | Sodium Oxide | Strongly basic |
| MgO | Magnesium Oxide | Basic |
| Al₂O₃ | Aluminium Oxide | Amphoteric |
| SiO₂ | Silicon oxide | Weakly acidic |
| P₂O | Phosphorous pentoxide | Acidic |
| SO₂ | Sulphur dioxide | Strongly acidic |
| Cl₂O7 | Chlorine heptoxide | Very strongly acidic |
On going down in a group of the periodic table, the order is reversed, i.e. basic character of oxides increases and acidic character of oxides decreases. e.g. in case of oxides of elements of group 2, the nature varies as:
| Oxide | Nature |
| MgO | Least basic |
| CaO | ↓ |
| SrO | |
| BaO | Most basic |
Electronegativity
It may be defined as the relative electron attracting tendency of an atom for a shared electron pair in a covalent bond with other atom.
Variation Along a Period and a Group
The electronegativity of the elements increases along a period since the non-metallic character increases. Similarly, it decreases down the group since the non-metallic character decreases.