STUDY NOTES of Metals And Non Metals
Metals – Physical Properties of Metals
Metals
Those elements which form positive ions by losing electrons are called metals. copper, iron, aluminium, sodium etc.
Physical Properties of Metals
(i) Metallic lustre : In pure state, metals have a bright shining surface. This property is called metallic lustre. Metals like gold, silver and platinum are known for their shining surface.
(ii) Hardness Most of the metals are hard : The hardness varies from metal to metal. Some alkali metals like lithium,
sodium and potassium are so soft that they can be easily cut with a knife.
(iii) Ductility: Metals are generally ductile. It is the property due to which a metal can be drawn into thin wires. Gold is the most ductile metal.
(iv) Malleability : Most of the metals are malleable. It is the property of metal due to which it can be beaten into thin sheets. Gold and silver are the most malleable metals.
(v) Electrical conductivity : Most of the metals are good conductors of electricity in solid state. However, conductivity may vary from one metal to another.The conduction of electricity or flow of electric current occurs due to the flow of free electrons present in the metal.
(vi) Good conductor of heat : Generally metals are good conductors of heat, except lead and mercury, which are poor conductors of heat. Metals like copper and silver are among the best conductors of heat.
(vii) Melting and boiling points : Metals generally have high melting and boiling points. Tungsten has the highest melting point among metals, while gallium and caesium have very low melting points. These two metals will melt if we keep them on our palm.
(viii) Sonority : The metals that produce a sound on striking a hard surface are said to be sonorous. Using this property, school bells are made up of metals.
Reaction of Metals with Oxygen (Burning in Air or Formation of Oxides)
Almost all metals combine with oxygen (or air) to form metal oxides.
Metal + Oxygen Metal oxide
For example, aluminium forms aluminium oxide, when heated in air.
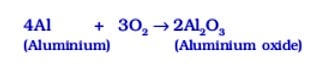 Similarly, when copper is heated in air, it combines with oxygen to form copper (II) oxide, a black oxide.
Similarly, when copper is heated in air, it combines with oxygen to form copper (II) oxide, a black oxide.

Generally, metal oxides are basic in nature.
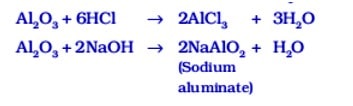
Exception Some metal oxides such as aluminium oxide, zinc oxide show both acidic and basic behaviour, such metal oxides which react with both acids as well as bases to produce salt and water are called amphoteric oxides.
Aluminium oxide reacts with acids and bases in the following manner:
Metallic oxides are insoluble in water but some of these dissolve in water to form hydroxides known as alkali.
sodium oxide and potassium oxide dissolve in water to produce alkalies as follows :
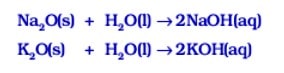
Order of Reactivity of Metals with Oxygen
Different metals react with oxygen at different rates, sodium (Na) and potassium (K) react so vigorously that oxygen catches fire if left in the open.
Hence, these are the most reactive metals. Therefore, to prevent accidental fires, these metals are kept immersed in kerosene oil.
At room temperature, the surfaces of magnesium and aluminium are covered with a thin layer of oxide which prevents the metal from further oxidation. Magnesium (Mg) and aluminium burns in air only by heating.
Zinc (Zn) burns only on strong heating while iron (Fe) does not burn in the form of rod or block but burns in the form of filing only. Copper (Cu) does not burn on heating but blister copper burns.
Silver and gold do not react with oxygen even at high temperatures. Hence, the order of reactivity of these metals with oxygen is
Na > Mg > Zn > Fe > Cu > Ag
Anodising
It is the process of forming a thick oxide layer on the surface of aluminium. Aluminium develops a thin layer of oxide, when left in the air. This oxide layer is protective and prevents the metal from further oxidation. This layer can be made more thick by anodising. In this process, clean aluminium articles are taken as anode and dil. I-1,SO4 as an electrolyte.
When electric current is passed, 02 gas gets liberated, which reacts with metal aluminium to form a thicker layer of oxide on its surface.
Reaction of Metals with Water
Metals react with water and produce a metal oxide and hydrogen gas. Metal oxides that are soluble in water dissolves in it further to form metal hydroxide. All metals do not react with water as the metals placed lower in the reactivity series are less reactive towards water.
Metal + Water Metal oxide + Hydrogen gas
Metal oxide + Water Metal hydroxide
(i) Metals like potassium and sodium react violently with cold water. In the case of sodium and potassium, the reaction is very violent and exothermic.

The heat evolved is sufficient for hydrogen to catch fire. That’s why, Na and K catch fire when kept in water. Therefore, both these metals are kept in `kerosene’ in order to avoid contact with both air and water.
(ii) The reaction of calcium with water is less violent. The heat evolved is not sufficient for the hydrogen to catch fire.
![]()
Calcium (Ca) floats over water because the bubbles of hydrogen gas formed stick on the surface of the metal.
(iii) Metals like aluminium, iron and zinc do not react either with cold or hot water. They react with steam and form the metal oxide and hydrogen.
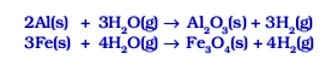
(iv) Lead, copper, silver and gold do not react with water at all. Thus, the reactivity order of metals toward water is
K > Na > Ca > Mg > > Fe > > Cu > Ag > Au
Reaction of Metals with Acids
(1) Reaction of metals with dil. HCl Except a few less reactive metals (such as Cu, Hg, Ag, Au, Pt etc), all metals react with dilute sulphuric acid and hydrochloric acid to produce salt and hydrogen gas.
Metal + Dilute acid –> Salt+ Hydrogen
![]()
(ii) Reaction of metals with dil. NO₃ Hydrogen gas is not evolved when a metal reacts with nitric acid. This is due to the strong oxidising nature _of nitric acid. It oxidises the H₂ produced to water and itself get reduced to any of the nitrogen oxide
(N₂0, NO, NO₂ ). But magnesium (Mg) and manganese (Mn) react with very dil. HNO₃ to evolve H₂ gas.
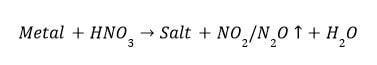
Exceptional case (Only for Mn and Mg)
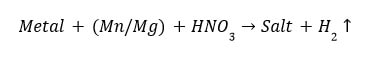
Aqua-regia (Latin for ‘royal water’)
It is a freshly prepared mixture of concentrated hydrochloric acid and concentrated nitric acid in the ratio of 3 : I. It can dissolve gold, even though neither oLgistse acid can do so alone. Aqua-regia is a highly corrosive, fuming liquid. It is one of the few reagents that is able to dissolve gold and platinum.
Reaction of Metals with Solutions of Other Metal Salts
Reactive metals can displace a comparatively less reactive metal from its compounds in an aqueous salt solution or in molten form. General equation is
![]()
This type of reaction is called displacement reaction.
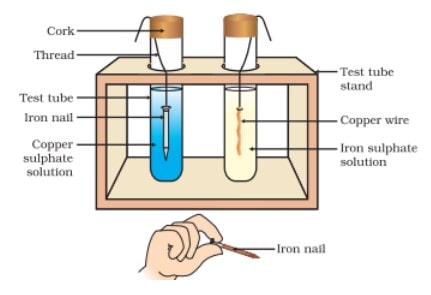
The Reactivity Series of Metals
The rectify series is a list of metals listed in increasing order of activity.
Metals are classified in a series based on their relative ability to lose electrons and their reactive nature; this series is known as the activity series or reactivity series of metals.
Most reactive metals(K, Na, Ca, etc.) are those that are put above hydrogen, whereas least reactive metals (noble metals, i.e gold and platinum ) are those that are placed below hydrogen.

Non-Metals
Non-metals are elements that obtain electrons from negative ions, such as iodine, sulphur, oxygen, and hydrogen. Except for bromine, which is a liquid, the non-metals are solids or gases.
Physical Properties of Non-metals
Non-metals aren’t malleable or ductile in the same way as metals are. They can’t be beaten into thin sheets or pulled into wires, in other words.
The nature of non-metals is that they are brittle. Sulphur, for example, is a brittle solid. It fractures into pieces when crushed.
The majority of non-metals are soft (if solid ). The hardest known material is diamond, an allotropic form of carbon.
Non-metals lack lustre, or a shining surface. Diamond, graphite (allotropic forms of carbon), and iodine, on the other hand, have shine despite being non-metals.
Although non-metals are poor conductors of carbon, they are excellent conductors of electricity.
Non-metals have low melting and boiling points in general. Nonmetals that are solids, on the other hand, have higher boiling points. Non-metals that are solids, such as B, Si, and others, have higher boiling points.
Chemical Properties of Non-metals
Non-metals do not react with water, own or dilute acid The reason is that they act as an elocution acceptor and cannot supply electrons to the 1-1 . ions of acids to taint them to hydrogen gas. But on heating, marimba inerra oxides or salts with cones. Acids.
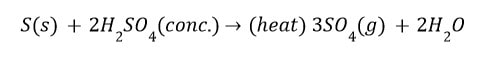
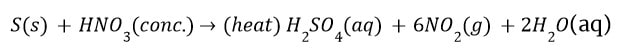
Reaction between Metals and Non-Metals (Ionic Bond Formation)
Each element wants to have a completely filled valence shell, it wants to have either 2 or 8 electrons in their outermost shell.
Metals have a tendency to lose electrons and non-metals have a tendency to gain electrons. When metals and non-metals react with each other then both of them try to achieve a completely filled outermost shell by the transfer of electrons.
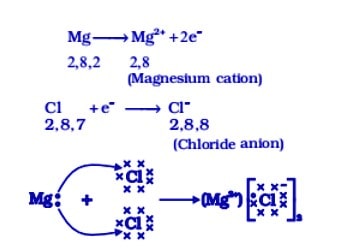
The electronic configuration of sodium (which is a metal) is K M L 2 8 1, and in order to complete its octet, ( 8 electrons in the outermost shell), it is easier for sodium to lose one electron from the M shell rather than accepting 7 electrons in it.
Thus, it has a tendency to lose one electron to have a completely filled valence shell. Similarly, if we see the electronic configuration of chlorine (configuration ,fg 1, a non-metal), we found that it is easier for chlorine to gain one electron in its M shell rather than losing 7 electrons from it. So, it has a tendency to gain electrons to have completely filled orbitals.
If sodium and chlorine react with each other then an electron lost by sodium (Na+ ) is gained by chlorine (Cl – ). Na + and Cl – ions being oppositely charged, attract each other and are held by strong electrostatic forces of attraction to exist as NaCl. Thus an ionic bond is formed between them.
Ionic or Electrovalent Bond
A chemical bond formed by the complete transfer of electrons from one atom to another is called an ionic bond. Such compounds are called ionic compounds.
A metal loses electron(s ) to form cations (+ ve ions).
A non-metal gains electrons to form anions (—ve ions). It means an ionic bond is formed between a metal and a Non-metal. Formation of ionic bonds can further be understood by taking an example of magnesium chloride (where magnesium is a metal and chlorine is a non-metal).
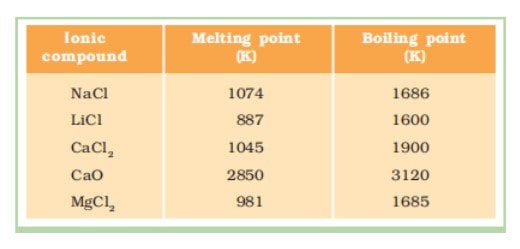
Properties of Ionic Compounds
(i) Physical nature : Ionic compounds are hard crystalline solids because of strong forces of attraction between the positive and negative ions. These compounds are generally brittle and break into pieces when pressure is applied.
(ii) Melting and boiling points : These compounds have high melting and boiling points as large amount of energy is required to break strong inter-ionic
attraction.
(iii) Solubility : These compounds are soluble in water (polar solvent) and insoluble in organic solvents (non-polar solvent) like kerosene, benzene, ether, petrol etc. As water is polar in nature it helps in separation of oppositely charged ions from their
ionic compound.
(iv) Conduction of electricity : The conduction of electricity through a solution involves movement of charged particles. Ionic or electrovalent compounds are good conductors of electricity, but they conduct electricity either in molten form or in their aqueous solution.
A solution of an ionic compound in water contain ions, which move to the oppositely charged electrode when electricity is passed through it. In molten form, the electrostatic forces of attraction between oppositely charged ions are overcome due to heat.
This is the reason due to which ions move freely and conduct electricity. They do not conduct electricity in solid form because movement of ions in
The solid state is not possible due to their rigid structure.
Occurrence of Metals
The earth’s crust is the major source of metals. Seawater also contains soluble salts like sodium chloride, magnesium chloride etc. The elements or compounds which occur naturally in the earth crust are known as minerals.
Those minerals from which metals can be extracted profitably are called ores.
Extraction of Metals
The process of obtaining pure metal from its ore is called extraction of metals. Some metals are found in earth’s crust in free state while some are found in the form of their compounds.
The metals present at the bottom of the reactivity series are least reactive, so they are found in free state, gold, silver, platinum, copper etc.
The metals at the top of the reactivity series (K, Na, Ca, Mg and Al) are highly reactive, so they are not found in nature as free elements.
The metals in the middle of the reactivity series (Zn, Fe, Pb, etc.) are moderately reactive and they are found as oxides, sulphides or carbonates in the earth’s crust. Ores of many metals are oxide, as oxygen is a very reactive element and is very abundant on the earth.
Thus, the different techniques used for extraction of metals depend upon their position in the activity series which is divided into three categories:
(i) Metals of low reactivity
(it) Metals of medium reactivity
(iii) Metals of high reactivity
Summary of several steps involved in the extraction of pure metal from their ores is given below in the form of flow chart followed by their detailed description:
Enrichment of Ores
The undesirable impurities like soil, sand, etc., found in ore are called gangue or matrix.Removal of gangue from the ore is called enrichment or concentration of ore.
The processes used for removing the gangue from an ore are based on the differences between the physical and chemical properties of the gangue and the ore. Depending upon the nature of impurities, different-separation techniques for enrichment of ores are employed.
Extraction of Metals (Present at the Bottom of the Activity Series) of Low Reactivity
These metals, being less reactive, can be obtained by reducing their
oxides to metals by heating alone.
(i) Cinnabar (HgS) It is an ore of mercury. When heated in air, it first changes into its oxide, HgO and then reduced into mercury metal on further heating.

(ii) Copper glance (Cu,S) When it is heated in air, partially oxidised and the oxidised product reacts with the remaining copper glance to give copper metal.

Extraction of Metals (Present at the Middle of the Activity Series) of Medium Reactivity
The metals in the middle of the activity series such as iron, zinc, lead, copper, etc., are moderately reactive. These metals are usually present as sulphides or carbonates in
nature. These sulphides or carbonates are first converted into oxides because it is easy to extract metals from its oxide.
Sulphides are converted into oxides by roasting and carbonates are converted into oxides by calcination. The metal oxides thus obtained are then reduced to the
corresponding metals by reduction using suitable reducing agents such as carbon.
Chemical reactions involved in the roasting and calcination of zinc ores are as follows:
Roasting It is the process in which a sulphide ore is heated below its melting point in the presence of excess air to convert it into metal oxide.
![]()
Calcination It is a process in which a carbonate ore is heated below its melting point in the absence of air to convert it into metal oxide.
![]()
Reduction of oxide ore It is the process of conversion of metal oxide ore into metal. It can be done by heating the oxides with suitable reducing agents like carbon in the form of coke.
![]()
Sometimes displacement reactions can also be used to reduce metal oxides to metals. The highly reactive metals such as sodium, calcium, aluminium etc., are used as reducing agents because they can displace metals of lower reactivity from their compounds,
Reaction of manganese dioxide with aluminium powder.
![]()
These displacement reactions are highly exothermic. The amount of heat produced is so high that the metals are produced in the molten state. The reaction of iron (III) oxide (Fe 203 ) with aluminium to produce iron is used to join railway tracks or cracked machine parts. This process is called thermite welding.
![]()
This reaction of metal oxide to form metal by using aluminium powder as a reducing agent is known as thermite reaction.
Extraction of Metals (At the Top of the Activity Series) of High Reactivity
The metallic compounds at the top of the activity series cannot be reduced by carbon or any other reducing agent due to their high affinity with oxygen. Therefore, electrolytic reduction is employed for metals like Na, Mg, Ca etc.
Electrolytic reduction The salts of these metals like chlorides in molten form are electrolysed. Metal is deposited at the cathode (the negative electrode) and chlorine is liberated at the anode (the positive electrode).
The reactions are as follows :
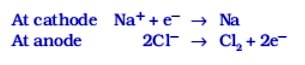
Similarly, aluminium is obtained by the electrolytic reduction of molten aluminium oxide also (called alumina).
Refining of Metals
It is the process of purification of the metal obtained after reduction. Various methods for refining are employed, but the most common one is electrolytic refining.
Electrolytic Refining
Many metals like Cu, Zn, Ni, Ag, Au etc., are refined electrolytically.
In this process, a thick block of impure metal is used as anode and a thin strip of pure metal is used as cathode. A solution of metal salt (to be refined) is used as an electrolyte.
When electric current is passed, metal ions from the electrolyte are reduced as metal which gets deposited on the cathode. An equivalent amount of pure metal from the anode gets oxidised to metal ion and goes into the electrolyte and from there it goes to cathode and deposit.
This cycle is repeated until the whole of the metal ion from the impure block is dissolved and deposited on cathode. The soluble impurities go into the solution, whereas the insoluble impurities settle down below anode and are known as anode mud, e.g. in electrolytic refining of crude copper.
Here, anode is impure copper whereas cathode is a strip of pure copper. The electrolyte is a solution of acidified CuSO 4 .On passing electric current, pure copper is deposited on the cathode.
Corrosion
It is the slow process of eating away of metals by the reaction of atmospheric air and moisture, e.g. rusting of iron, tarnishing of silver, formation of green coating over copper etc.
Prevention of Corrosion
Rusting of iron is prevented by galvanising, by making alloys, painting, greasing or oiling and tin-plating and chrome plating (chromium plating) which are explained
below :
- Galvanisation The process of coating iron and steel objects with a thin layer of zinc is called galvanisation. It is done by dipping the object in molten zinc. The galvanised article is protected against rusting even if the zinc coating is broken.
- Alloying It is the method of improving the properties of a metal by mixing the metal with another metal or non-metal.
Alloy
A homogeneous mixture of two or more metals or a metal and a nonmetal is defined as an alloy. It is made by combining molten metals and then cooling the mixture.
An alloy’s electrical conductivity and melting point are lower than those of pure metals; for example, brass, an alloy of copper and zinc (Cu and Zn), and bronze, an alloy of copper and tin (Cu and Sn), are not good conductors of electricity, whereas copper is used to make electrical circuits.
Solder, a lead-tin alloy, has a low melting point and is used to join electrical wires. If an alloy contains mercury as one of its constituents, it is referred to as amalgam, for example, sodium-mercury amalgam, silver-mercury amalgam, and so on.
Alloying of gold
Pure gold is very soft. It is called 24 carat gold. To increase the strength and hardness of gold and to make it suitable for making jewellery, alloy of gold is made either with silver or copper. e.g. 22 carat gold means 22 parts pure gold mixed with 2 parts of Cu or Ag.
Alloying of iron
Pure iron is very soft and stretches easily when hot. It is mixed with a small amount of carbon (about 0.05%) and it becomes hard and strong. Iron is mixed with many metals
to form different alloys. For e.g. when iron is mixed with nickel and chromium. stainless steel is obtained, which is hard and does not rust.
- Painting : Rusting of iron can be easily prevented by coating the surface with a paint which protects iron from air and moisture.
- Greasing or Oiling : When grease or oil is applied to the surface of an iron object then air and moisture cannot come in contact with it and hence, rust is prevented, e.g. tools and machine parts made up of iron are smeared with grease.
- Tin Plating and Chromium: Plating Tin and chromium metals are resistant to corrosion. So, when a thin layer of tin metal is deposited on an iron object by electroplating, then iron and steel objects are protected from rusting.
The Wonder of Ancient Indian Metallurgy
The iron workers of India built the Iron Pillar near Qutub Minar in Delhi over 1 600 years ago. They had invented a method to keep iron from rusting. This is because they have a thin layer of magnetic oxide, Fe 304, on their surface. It has been studied by scientists from all over the world for its rust resistance. The Iron Pillar stands 8 metres tall and weighs 6 tonnes (6000kg).
NCERT Notes for Class 10 Science Chapter 3 Metals and Non-metals
Question 1. Give example of a metal which
(a) is a liquid at room temperature
(b) can be easily cut with a knife
(c) is the best conductor of heat
(d) is the poorest conductor of heat.
Answer:
(a) Mercury
(b) Sodium
(c) Silver
(d) Lead.
Question 2. Explain the meaning of malleable and ductile.
Answer:
Malleable: The ability of a material to be beaten into thin sheets is called its malleability. Metals can be bent or shaped.
Ductile. The ability of a material to be pulled into wires is called its “ductility.” Metals are ductile in nature.
Question 3. Why is sodium kept immersed in kerosene oil ? (CBSE 2011)
Answer: Both air and water can change sodium. It is a metal that reacts quickly. When left out in the open, it mixes easily with the oxygen in the air to make its oxide. In the same way, it forms sodium hydroxide when it comes into contact with water vapours or moisture.
Sodium metal is usually kept under kerosene so that neither air nor water can get to it.
Question 4. Write the equations for the reactions of
(a) iron with steam
(b) calcium with water
(c) potassium with water.
Answer:
(a) 3Fe(s) + 4H2O (steam) ———–> Fe3O4(s) + 4H2(g)
(b) Ca(s) + 2H2O(aq) ———–> Ca(OH)2(s) +H2(g)
(c) 2K(s) + 2H2O(aq) ———–> 2KOH(aq) + H2(g)
Question 5. Samples of four metals A, B, C and D were taken and were added to the following solutions one by one. The results obtained have been tabulated as follows :
| Metal | Solution to which metal is added | |||
| Iron(II) sulphate | Copper(II) sulphate | Zinc sulphate | Silver nitrate | |
| A | No reaction | Displacement | — | — |
| B | Displacement | — | No reaction „ | — |
| C | No reaction | No reaction | No reaction | Displacement |
| D | No reaction | No reaction | No reaction | No reaction |
Use the table given above to answer the following questions :
(a) Which is the most reactive metal ?
(b) What would you observe when B is added to solution of copper(II) sulphate ?
(c) Arrange the metals A, B, C and D in order of increasing reactivity. (CBSE 2011)
Answer:
Based on the activity series, the relative position of the metals in solutions is : Zn > Fe > Cu > Ag. On the basis of the results given in the table .
- Metal A is more reactive than copper and less reactive than iron.
- Metal B is more reactive than iron and less reactive than zinc. –
- Metal C is only more reactive than silver and less reactive than other metals.
- Metal D is the least reactive in nature.
In the light of above information, we can conclude that
(a) Metal B is the most reactive.
(b) B is more reactive than iron and copper because iron is more reactive than B. This means that it would take the place of copper in a solution of copper(II) sulphate. Solution will slowly lose its blue colour.
(c) The decreasing order of reactivity of metals is: B>A>C>D.
Question 6. Which gas is produced when a reactive metal reacts with dilute hydrochloric acid ? Write the chemical reaction when iron reacts with dilute H2SO4. (CBSE 2010)
Answer: Hydrogen gas (H2) is produced when a reactive metal reacts with dilute hydrochloric acid. Iron and dilute H2SO4 react as follows :
Fe(s) + H2SO4(aq) ————> FeSO4(aq) + H2(g)
Hydrogen gas is evolved in this reaction also.
Question 7. What would you observe when zinc is added to a solution of iron (II) sulphate ? Write the chemical reaction that takes place. (CBSE 2010)
Answer: The solution’s green colour would slowly go away. Zinc would slowly break down, while iron would settle to the bottom of the beaker.
Question 8.
(i) Write electron-dot structures for sodium, magnesium and oxygen.
(ii) Show the formation of Na2O and MgO by the transfer of electrons.
(iii) What are the ions present in these compounds ?
Answer:
- Formation of sodium oxide (Na2O)
- Formation of magnesium oxide (MgO)
- For answer, consult structures given above.
Question 9.Why do ionic compounds have high melting points ? (CBSE 2014)
Answer: Ionic compounds are made up of positive ions (called cations) and negative ions (called anions). Ionic compounds are crystalline solids that are tightly packed. They have strong forces of attraction between their ions, and their melting and boiling points are high.
Question 10. Define the following terms
- Minerals
- Ores
- Gangue.
Answer:
Minerals: This is how metals and non-metals in the earth’s crust are mixed together.
Ores are the minerals that can be used to easily and profitably get metals out of them.
Gangue: It stands for the mud, sand, and clay that are mixed in with the ore and make it less pure.
Question 11.Name two metals which are formed in nature in free state.
Answer: The metals are gold (Au) and platinum (Pt).
Question 12.Which chemical process is used for obtaining a metal from its oxide ?
Answer: The chemical process is known as reduction.
Question 13.Metallic oxides of zinc, magnesium and copper were heated with the following metals. In which cases, will you find displacement reactions taking place ?
| Metal | Zinc | Magnesium | Copper |
| Zinc oxide Magnesium oxide Copper oxide |
Relative positions of these metals in the activity series are : Mg, Zn, Cu : In the light of this :
Answer: Magnesium (Mg) will displace both zinc (Zn) and copper (Cu) from their oxides
Mg + ZnO ———-> MgO + Zn
Mg + CuO ———-> MgO + Cu
Zinc will displace copper from copper oxide.
Zn + CuO ———–> ZnO + Cu
Copper is least reactive and will not initiate displacement reaction.
Question 14.Which metals do not corrode easily ?
Answer: Metals at the bottom of the activity series, like gold (Au) and platinum (Pt), don’t corrode easily.
Question 15.
What are alloys ? (CBSE 2011)
Answer: Alloys are the homogeneous mixture of two or more metals or even metals and non-metals.
Question 1.Which of the following will give displacement reactions ?
(a) NaCl solution and copper metal
(b) MgCl2 solution and aluminium metal
(c) FeSO4 solution and silver metal
(d) AgNO3 solution and copper metal.
Answer: (d). Only the AgNO3 solution will give displacement reaction with copper (Cu) because copper is placed above silver in the activity series.
Question 2.Which of the following methods is suitable for preventing an iron frying pan from rusting ?
(a) applying grease
(b) applying paint
(c) applying a coating of zinc
(d) all the above.
Answer: In theory, all three of these ways can help keep an iron frying pan from rusting. But most of the things that make up grease and paint are organic compounds. They can’t handle the heat, so they don’t last. So, the best way is to use galvanisation, which is a zinc coating. Choice (c) is right.
Question 3. An element reacts with oxygen to give a compound with high melting point. This compound is also water soluble. The element is likely to be :
(a) Calcium
(b) Carbon
(c) Silicon
(d) Iron
Answer: (a). Calcium (Ca) combines with oxygen to form calcium oxide (CaO) with a very high melting point. CaO dissolves in water to form calcium hydroxide
Question 4.Food cans are coated with tin and not with zinc because
(a) Zinc is costlier than tin
(b) Zinc has higher melting point than tin
(c) Zinc is more reactive than tin
(d) Zinc is less reactive than tin.
Answer: (c). Zinc is more reactive than tin, and when it mixes with organic acids in food, it makes compounds that are poisonous. Tin is less reactive than zinc because it is below it on the activity scale. It does not react with organic acids. This means that (c) is the right answer.
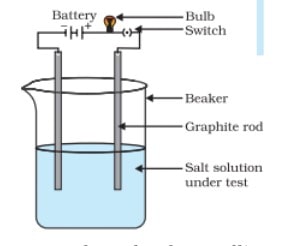
Question 5.You are given a hammer, a battery, a bulb, wires and a switch:
(a) Flow could you use them to distinguish between samples of metals and non metals ?
(b) Assess the usefulness of these tests to distinguish between metals and non metals.
Answer:
(a) Use a hammer to turn both the solid metal and solid non-metal into plates or rods. Since metal is easy to shape, it will be easy to make these. If you hit a non-metal with a hammer, it will break because it is not strong. It will be hard for them to make plates.
Now, use these plates as electrodes to build a cell in each case and turn on the current. If the light bulb glows, then the electrodes are made of metal. If this doesn’t light up, it means that the electrodes are made of something other than metal.
(b)Based on these tests, we could say that metals can be shaped but non-metals can’t.
Metals are good at letting electricity flow through them, but non-metals are not (graphite is an exception).
Question 6. What are amphoteric oxides ? Give examples of two amphoteric oxides.
Answer: These are the oxides which can act both as acids and bases. For example, aluminium oxide (Al2O3) and zinc oxide (ZnO). The amphoteric character of the two oxides are shown by the following reactions.
Question 7. Name two metals which can displace hydrogen from dilute acids and two metals which can not do so.
Answer: Sodium and calcium can displace hydrogen from dilute acids Copper and silver can’t take hydrogen out of weak acids.
Question 8. In the electrolytic refining of metal M, name anode, cathode and electrolyte.
Answer:
Anode : Rod of the impure metal
Cathode : Rod of pure metal
Electrolyte : Aqueous solution of soluble salt of metal M.
Question 9.Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it as shown in the figure.
What will be the action of gas on
- dry litmus paper ?
- moist litmus paper ?
Write a balanced chemical equation for the reaction taking place.
(CBSE 2011)
Answer: The gas evolved upon heating sulphur powder on a spatula is sulphur dioxide
- SO2(g) has no action of dry litmus paper.
- SO2(g) dissolves in moisture (water) present in moist litmus paper to form sulphurous acid. In acidic solution, moist litmus paper will change to red.
Question 10.State two ways to prevent the rusting of iron.
Answer:
- By putting a layer of grease or paint on the iron’s surface.
- By adding a layer of zinc to the iron’s surface. The name for this process is galvanisation.
Question 11.What types of oxides are formed when non-metals combine with oxygen ?
Answer: The oxides are generally acidic in nature which means that when dissolved in water, their solutions change blue litmus to red. For example,
Question 12. Give reasons for the following :
(a) Platinum, gold and silver are used to make jewellery.
(b) Sodium, potassium and lithium are stored under oil.
(c) Aluminium is a highly reactive metal but still used for making cooking utensils.
(d) Carbonate and sulphide ores are usually converted into oxides during the process of extraction. (CBSE 2013, 2014)
Answer:
(a) These metals, which are at the bottom of the activity scale, don’t react much in nature. Noble metals include gold, platinum, and palladium.
They aren’t affected by air, water, or chemicals. These metals can be used to make jewellery because they shine brightly.
(b) On the activity scale, there are metals that react easily. Because oxygen, water vapour, and carbon dioxide are in the air, their surface gets dirty.
When put in water, these react violently and give off so much heat that they can’t be handled. Most of the time, these metals are kept under kerosene, which doesn’t have any air or water in it.
(c) When the metal is exposed to air, it changes into aluminium oxide (Al2CO3). It sticks to the metal’s surface and makes a coating that protects the metal.
Because this layer is there, the metal is no longer reactive and can be used to make cooking tools.
(d) Both carbonate and sulphide ores of metals can’t be directly reduced to the metal state. Metal oxides, on the other hand, are easy to get rid of using coke or other reducing agents.
The calcination process (for carbonate ores) and the roasting process change both into their respective oxides (for sulphide ores).
With coke (C) or another suitable reducing agent, it is easy to turn metal oxides back into metal. So, carbonates and sulphides are not directly reduced. Instead, calcination and roasting are used to change them into oxides.
Question 13. You must have seen tarnished copper vessels being cleaned with lemon or tamarind juice. Explain why these sour substances are effective in cleaning the vessels. (CBSE 2014)
Answer:
Copper metal reacts slowly with water, carbon dioxide, and oxygen in the air to make green basic copper carbonate. Its layer slowly builds up on the metal’s surface..
Now, lemon juice contains citric acid, while tamarind has tartaric acid. Both of these acids react with basic copper carbonate to form soluble salts like copper acetate (with citric acid) and copper tartrate (with tartaric acid).
The equations for the reactions are complicated and aren’t given. When these salts are taken off the surface of the copper metal, the surface of the metal shines.
Question 14.A man went door to door posing as a goldsmith. He promised to bring back the glitter on dull gold ornaments. An unsuspecting lady gave a set of gold bangles to him which he dipped in a particular solution.
The bangles sparked like new but their weight was reduced drastically. The lady got upset and after a futile argument, the man beat a hasty retreat. Can you predict the nature of the solution used by the imposter ?
Answer:
The man had actually used a solution of aqua regia, which is a mixture of cone.HCl and cone. HNO₃ in the proportion of 3:1 by volume. This solution dissolved gold into soluble auric chloride (AuCl3).
Because the gold did react, the weight of the gold bangles went down. When the dull layer of gold on the surface was taken off, the bangles’ original shine came back. There have been chemical reactions.
Question 15. Give reason as to why copper is used to make hot water tanks and not steel (an alloy of iron).
Answer:
Steel, which is an iron alloy, moves heat better than copper. Even though copper costs more than steel, it is used instead of steel to make hot water tanks.
Question 16.Differentiate between metals and non-metals on the basis of chemical properties.
For the distinction in the chemical characteristics,
Metals :
- Nature of oxides : Most metals’ oxides are basic in nature. (Exception : ZnO and Al2O3 are amphoteric oxides)
- Electrochemical behaviour : Most of the time, metals become cations when they lose electrons. This means that they have an electric charge.
- Action with dilute acids : Active metals evolve hydrogen on reacting with dilute HCl and dilute H2SO4
- Nature of compounds : Most of the time, metal compounds are ionic.
- Oxidising and reducing nature : Metals act as reducing agents as their atoms lose electrons. For example,
Na —— > Na+ + e–
Non-metals :
- Nature of oxides : Most of the time, non-metal oxides are acidic in nature. (Exception : CO and N2O are neutral oxides)
- Electrochemical behaviour : When non-metals gain electrons, they usually make anions. This means that they have a negative charge.
- Action with dilute acids : Dilute acids do not react with non-metals.
- Nature of compounds : Most non-metal compounds are covalent, but there are a lot of exceptions.
- Oxidising and reducing nature : Because their atoms can take on electrons, non-metals are oxidising agents. One example is
Cl + e–——–>Cl–